
IPAC GTA Product Showcase
VanishPoint
VanishPoint® syringes are available in a variety of sizes, needle gauges, and needle lengths. The needle is automatically retracted directly from the patient into the barrel of the syringe when the plunger handle is fully depressed. The pre-removal, automated retraction virtually eliminates exposure to the contaminated needle, effectively reducing the risk of needlestick injury.
EasyPoint
EasyPoint retractable needles feature automated retraction technology and pre-removal capability that has made Retractable Technologies the industry standard, but with added versatility. They can be used with luer lock and luer slip syringes, including pre-filled syringes. When the color-coded activation tab is pressed, the needle automatically retracts into the safety chamber and remains enclosed through disposal. Upon needle retraction, the syringe luer tip is covered, reducing the risk of exposure to medication or blood and body fluids. EasyPoint retractable needles are easy to use, allowing single-handed activation, while the clinician’s fingers remain behind the exposed sharp at all times.
SecurePortIV
The only cyanoacrylate glue designed to secure all your venous catheters (peripheral catheters, Midline, PICC, CVC). Limits bleeding and catheter movement and forms a barrier against infection and moisture.

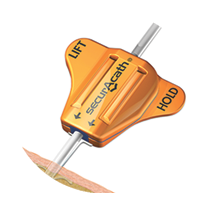
SecurAcath
Reduces CLABSI, prevents catheter dislodgement for the Life of the Line. Improves efficiency with catheter care & maintenance, significantly reduces nursing time compared to adhesive securement devices. Please visit our SecurAcath product page for more information.
CathGrip®
CathGrip® is the only true universal catheter securement system. The device is made from our proprietary hydrocolloid that is hypoallergenic, latex free, and moves like a second skin with wear time of up to 7 days! Our skin-friendly adhesive helps prevent skin tearing and irritation unlike other acrylic based securement devices. Other products that may claim to be hydrocolloid are actually only partially hydrocolloid while the CathGrip® is 100% hydrocolloid and therefore a much gentler application.

FreeDerm® Adhesive Removal
FreeDerm adhesive remover is our trademark adhesive remover. Specially formulated to instantly remove hydrocolloid tapes, and bandages, FreeDerm is available in spray, ampoule, or wipe form. The FreeDerm spray bottle is ergonomically designed for customers with limited manual dexterity. FreeDerm adhesive remover is for external use only. FreeDerm can be used anywhere on the body. The formula is quick-drying and does not require washing. It is environmentally friendly and does not contain ozone-depleting chloro-fluorocarbons.

BioPlus+™
BioPlus+™ skin prep wipes provide a barrier lm layer directly on the skin to improve the adhesion of hydrocolloids, appliances, tapes, and bandages. The wipes protect skin from incontinence/ body fluids and adhesive stripping. They are no-sting, Isopropyl alcohol-free, and dry quickly to form a transparent coating on the skin.

Lyka Ports
Uniquely designed for use in hemodialysis, the Lyka™ by Quest Medical1 brings engineering excellence to patient care, safety, and comfort.
The needleless connector utilizes the Quest split septum with low dead space and is fully flushable to reduce infection risk. With an open fluid path, it provides Ultra High flow rates ideal for your patient’s needs. The closed nature of the valve offers protection to the patient against catheter-related bloodstream infections (CRBSIs) and helps reduce clinical costs related to CRBSIs

FlowArt
Needle-free Hemodialysis Connector
- Flowrates >600 ml/min
- Straight Fluid Path
- Compatible with Standard Swabbing Cap
- Low Profile Design

Tristel ULT High Level Disinfectant foam for Ultrasound Probes
Tristel ULT is a high-level disinfectant foam with dry wipes intended to disinfect cleaned, reusable, non-lumened ultrasound probes. Used by professionals in 35 countries since 2008, Tristel ULT is now FDA-approved for sale in the United States. Tristel ULT is not dangerous for shipment and can be transported by air, road and sea without restrictions.

UltraDrape II
UltraDrape II is an evolutionary dual-action barrier and securement dressing for use during Ultrasound-Guided Peripheral Intravenous (UGPIV/USGPIV) procedures with the benefits of UltraDrape, plus a new size, improved “standability”, and seamless scanning from dressing to skin.
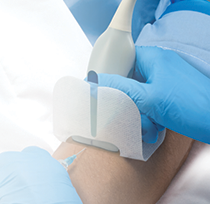
Punctur Guard
CT rated Positive pressure Huber needle, eliminates heparin flushing, and reduces TPA (alteplase) usage. Please visit our Punctur Guard product page for more information.

Safetouch II
Once locked, it stays locked!
The only system with built-in safety advantages: a permanent locking feature eliminates the worry of accidental needlesticks; textured wings provide secure grip; system is low profile and is designed to meet all OSHA safety requirements.
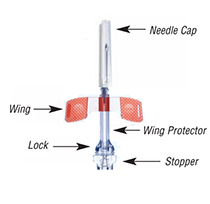
Tulip Safety Fistula Needle
Features of the clear, patented TULIP safety needle protector:
- Low profile design
- Ease of use during patient cannulation and needle placement
- Activation using a one-handed technique
- Audible click indicating protector is locked and will stay locked
- Meets OSHA safety requirements
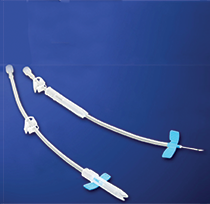
Supercath Neo
- Supercath Neo, Safety Fistula Catheter, helps to reduce accidental
needlesticks with passive and fully-encased needle shield - Applicable to AV fistula and AV graft
- Clamping the soft tube section with fingers or forceps enables connection to
blood line system without blood leakage - Side holes on catheter allow optimal and smooth blood flow
- Single piece jointless catheter design prevents clotting and ensures a smooth flow

Medikit SuperCath PRO
- Passive safety cover with fully-encased needle protection
- Built-in check valve that prevents the out Flow of blood andallows the blood line connection without clamping
- A gentle hemodialysis treatment with a plastic catheter tube
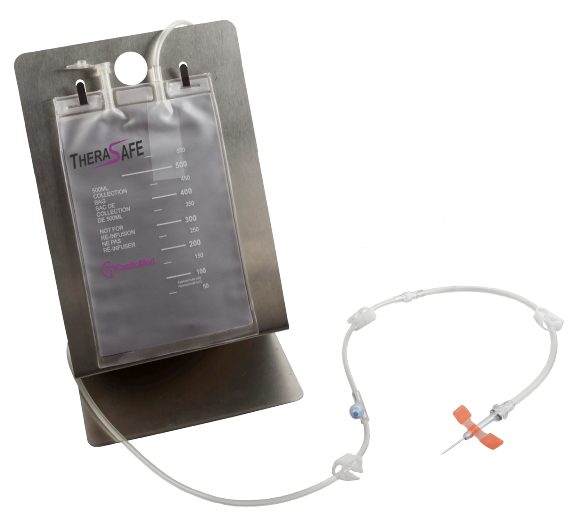
TheraSafe
Traditional methods for performing the therapeutic phlebotomy procedure utilized 500ml glass vacuum bottles, blood bags, or modified blood bags, each method having its own clinical precautions. THERASAFE is the only FDA-approved device specifically designed for Therapeutic Phlebotomy procedures.
CONTACT A SALES REPRESENTATIVE IN YOUR AREA
Bill Kroesen
Territory Account Manager: Western Canada
Contact Bill Kroesen
Please feel free to contact Bill Kroesen directly by phone at:
(604)-889-3721
or by email clicking the button below.
Amy Ludwig MSc.
Territory Account Manager: Manitoba Saskatchewan
Contact Amy Ludwig
(647) 981-0196
or by email clicking the button below.
Justin Dubé
Territory Account Manager: Eastern Ontario & Bermuda
Contact Justin Dube
Please feel free to contact Justin Dube directly by phone at:
(647) 567-6833
or by email clicking the button below.
Todd Townend
Territory Account Manager: Ontario
Contact Todd Townend
Please feel free to contact Todd Townend directly by phone at:
(647) 981-0196
or by email clicking the button below.
Christine Dubé
Territory Manager
Contact Christine Dubé
Please feel free to contact Christine Dubé directly by phone at:
(647) 459-7629
or by email clicking the button below.
Michel Paris
Représentant (N & E Québec, Maritimes)
Contact Michel Paris
Service bilingue: français / anglais
Please feel free to contact Michel Paris directly by phone at:
(581) 888-1358
or by email clicking the button below.
Elie Raie
Territory Account Manager: Eastern Canada
Contact Elie Raie
Service bilingue: français / anglais
Please feel free to contact Elie Raie directly by phone at:
(514) 949-0610
or by email clicking the button below.
CardioMed Supplies Inc.
199 St. David Street
Lindsay, ON, Canada
K9V-5K7
Phone: (705) 328-2518
Fax: (705) 328-9747
Toll-Free: 1-800-387-9757
Email: mail@cardiomed.com
Nipro Canada
199 St. David Street
Lindsay, ON, Canada
K9V-5K7
Phone: (705) 328-2518
Fax: (705) 328-9747
Toll-Free: 1-800-387-9757
Email: service@nipro.ca
